The Seasonal Influenza Virus Vaccine
It’s flu season again. And you have a decision to make. Should you get the flu vaccine this year?
Before you get started educating yourself on the influenza vaccine (and I applaud you for doing so), you may want to read the previous two articles in this flu education series:
- So many people are confused about what influenza really is because the word “flu” is often misused and confused for various other sicknesses going on in the body. To familiarize yourself with the true flu symptoms and to understand why you should get vaccinated, please read: What is influenza, “the flu?”
- Understanding a little more about how the influenza virus is constructed and how it mutates is also important in grasping why we all need to get the flu shot every year. I think you’ll be interested in reading more about the virus that causes the flu and learning: Why do we keep getting the flu over and over again?
Now that you’ve become flu savvy and have decided you are ready to make the choice to look into getting the vaccine, it’s important to know all you can before deciding which vaccine may be best for you and your loved ones.
First off, there are lots of different options when it comes to the flu vaccine. There are actually 13 different seasonal flu vaccine options out there this year (2014-2015) alone. For a full list of all the vaccine choices for the 2014-2015 flu season, the CDC has one available, here. But, before you read the list and get confused, let me break it down for you.
You are now able to get a vaccine with either three strains of the flu (trivalent), containing one influenza A H1N1 virus, an influenza A H3N2 virus, and one influenza B virus OR a vaccine with four strains of the flu (quadrivalent), containing the same three above viruses and one extra influenza B virus.
For decades we only had the trivalent version of the flu vaccine, which protected us from only one strain of influenza virus B. And often times, the strain picked for the following flu year was not the strain that ended up circulating. Now, there are two strain B’s in the quadrivalent vaccine, giving us much more coverage.
So which is better, the trivalent or the quadrivalent?
Safety wise, they are very similar. But, the quadrivalent is more expensive and may be harder to find. However, it offers a bit more protection during the flu season.
The U.S. Centers for Disease Control and Prevention (CDC) doesn’t recommend one over the other. And the major reason the CDC isn’t stating they favor one vaccine over the other is because they don’t want people to miss getting the flu vaccine just because they are waiting to find the quadrivalent version. There are often more limited quantities of the quadrivalent vaccine, and getting the trivalent vaccine is much safer than actually getting the flu.
According to the CDC, of the 151 to 156 million doses of influenza vaccines projected to be available for the 2014-2015 season, manufacturers estimate that 76 million doses will be quadrivalent flu vaccine.1 However, the quadrivalent vaccine may become more readily available in the future.
So if you are unable to find the quadrivalent version, go ahead and protect yourself by getting the trivalent vaccine.
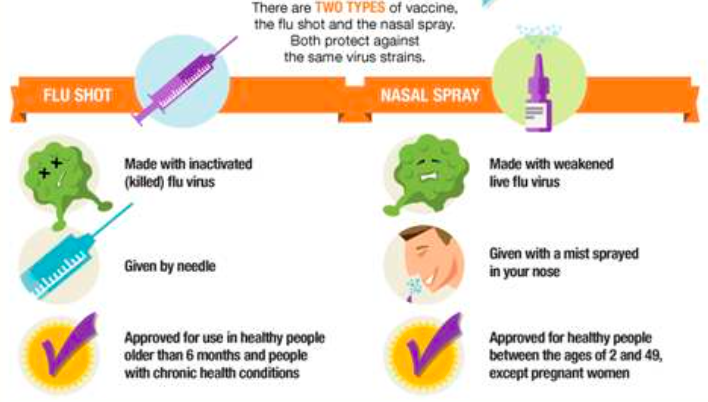
Now, the quadrivalent vaccine is available as the flu shot (an intramuscular shot usually given in the upper arm), or as a nasal spray. The trivalent version is only available as a shot. All of the shots contain inactivated (killed) flu particles particles, whereas the nasal spray contains weakened live viral particles. Either way, no live flu virus is ever part of this vaccine and can therefore NOT make you sick with influenza.
The live, yet weakened flu particles make up the nasal (FluMist) spray, which is sprayed into the nostrils. The nasal spray is approved for the use in healthy people 2-49 years old (and who are not pregnant). And starting this year (2014-2015), the CDC is recommending the use of the nasal spray vaccine for children 2-8 years old, as long as the child is otherwise healthy. However, if the nasal spray is not available, the CDC does not recommend waiting until it becomes available. The flu vaccine should not be delayed.
So, who shouldn’t get the flu vaccine?
You should not get the flu vaccine if:
- You have had a severe allergy to a previous dose of the flu vaccine, or have an allergy to gelatine, some antibiotics, or egg. (Most flu vaccines contain a small amount of egg protein.)
- You have ever had Guillain-Barré Syndrome discuss getting the vaccine with the doctor first.
- You are not feeling well you may be advised to wait to get the flu vaccine until you are feeling better.
You would be advised not to get the live nasal spray if:
- You have any of the above conditions
- You are under two years old or over 50 years old.
- You are a are under five with a history of wheezing.
- You have a medical condition that may increase your risk for flu complications, such as chronic heart or lung disease, diabetes or kidney failure, and people with illnesses or who are taking medication that may weaken the immune system.
- You are pregnant
- You are under 18 and receiving aspirin therapy
- You have allergies to gentamycin (an antibiotic) or arginine
Who definitely needs to get the flu vaccine?
- Young children
- People over 65 years
- Pregnant women
- People with heart, lung or kidney disease, nervous system disorders, or a weakened immune system.
- Anyone in close contact with someone in one of these categories
You should get the flu SHOT instead of the live nasal spray if:
- You are pregnant
- You have a weakened immune system
- You are a young child with asthma or wheezing problems
- You are a child or adolescent on long-term aspirin therapy
- You will provide care for, or visit someone, within the next 7 days who needs special care for an extremely weakened immune system
- You have taken influenza antiviral medications in the past 48 hours
These above mentioned persons, as well as health care workers, are considered high priority because they are considered at high risk for having complications from the flu . So should there be limited numbers of flu vaccine available, these groups of people will be given first access to the vaccine.
Some children 6 months through 8 years of age might need two doses during one year. Children getting vaccinated against the flu for the first time and some children who have had the vaccine previously will need two doses. The first dose will prime the immune system and should be given as soon at the vaccine becomes available. The second dose will offer protection from the strains of flu contained in the vaccine. If your child needs a second dose, be sure to get it. For more information, there is a good on at the CDC’s website and please talk to your child’s doctor about whether or not your child needs a second dose.
Another option for the flu vaccine is the trivalent intradermal shot. This vaccine goes right under the skin instead of into the muscle, and therefore uses a much smaller needle. It has been described as far less painful that the intramuscular shot. This contains the same trivalent strains as the flu shot does, and is only recommended for people 18-64 years. It may be a good option for those persons who have an extreme fear of needles.
In addition, there is also a high-dose flu shot available, and is recommended for persons over the age of 65. This contains four times more flu antigens, which may stimulate a greater immune response. Because the immune system weakens with age, people with advanced age are more susceptible to complications from getting the flu. Also, aging decreases the body’s ability to make proper antibodies, therefore injecting more antigens has been shown to give an aging body a greater immune response (higher levels of antibodies).
The safety profile for both the high dose vaccine and the intradermal vaccine is similar to that of the regular flu vaccine, which leads to is the flu vaccine safe?
The flu vaccines are safe. Trivalent flu vaccines have been around for many, many years, and have been shown to have a similar safety profile to the quadrivalent vaccine. (Adding one more flu strain doesn’t seem to have any serious side effects compared to the trivalent vaccine.)
Both the quadrivalent and the trivalent vaccines are unable to cause influenza illness.
All flu vaccines have been shown to be safe to be given at the same time as other vaccines. And even if you have a mild illness at the time of vaccination, it’s still usually okay to get the flu vaccine.
Some of the inactivated flu vaccines contain a small amount of thimerosal. However, studies show that this mercury-based preservative is not harmful. If you are worried, there are flu vaccines that do not contain any preservatives. Just ask your doctor for a preservative-free option.
What are the potential side effects with the flu vaccine?
Most of the potential side effects are mild and go away on their own. These side effects tend to be infrequent and minimal. But, it’s important to note that this vaccine, like all vaccines are medicines and there is a chance of side effects.
Some problems that can occur after getting ANY vaccine include:
- Rarely severe pain or reduced motion in the location the shot was given.
- Brief fainting spells after the procedure. Tell your doctor if you feel dizzy, have ringing in the ears, or have blurred vision.
- Severe allergic reaction (estimated less that 1 in 1 million doses)1, which would likely occur minutes to a few hours after vaccination. What to do if you suspect an allergic reaction.
Mild problems following inactivated flu shot:
- soreness, redness, or swelling where the shot was given (Less than about 1/3 of people develop soreness and accessional redness at the injection site)4
- hoarseness
- sore, red or itchy eyes
- cough
- fever
- aches
- headache
- itching
- fatigue
Mild problems following the live flu nasal spray:
- Children and adolescents 2-17 years of age:
- runny nose, nasal congestion or cough
- fever
- headache and muscle aches
- wheezing
- abdominal pain or occasional vomiting or diarrhea
- Adults 18-49 years of age:
- runny nose or nasal congestion
- sore throat
- cough, chills, tiredness/weakness
- headache
Some young children who are given the flu shot at the same time they are given the pneumococcal vaccine (PCV13) may be at a higher risk for febrile seizures (seizures caused by high fevers).
If these problems occur, they usually begin soon after the shot and last 1 or 2 days. About 5-10% of people experience mild side effects for about a day after vaccination.4 Most of these side effects occur in children who have not had the flu vaccine in the past. As with any medicine, there is a very remote chance of a vaccine causing a serious injury or death.
Some of these mild side effects may mimic symptoms of the flu and may lead people to believe they have contracted the flu from the vaccine. All of the flu vaccines do NOT contain live virus so getting the vaccine dose not cause disease.
It takes about 2-3 weeks for the body to develop protective antibodies to the antigens in the vaccine. So, you may not be covered until several weeks after you get the vaccine. The protection from the vaccine lasts for several months up to a year and should offer protection for the several months of the peak flu season.
The effectiveness of the flu vaccine is judged by its ability to generate a proper immune response (make sufficient antibodies), but the efficacy can vary from person to person. Studies show that in healthy young adults the flu vaccine proves to be between 70-90% effective in preventing influenza.4 In those who are elderly or have a chronic medical condition, the vaccine may be less effective due to the immune system’s inability to create a proper immune response.
Studies show that the vaccine reduces hospitalization rates by about 70% and death rates by about 85% among the elder who are not in nursing homes.4 For those in nursing homes the risk of hospitalization is decreased by about 50%, the risk of pneumonia by about 60%, and the risk of death by 75%-80%.4 The nasal spray has been found to be less effective in the older population, as well.4
The effectiveness as well as the safety of all vaccines are monitored by the FDA, CDC, and other institutions in the U.S. This monitoring is always ongoing and new vaccines such as the quadrivalent flu vaccine are being monitored very closely.
Because the influenza virus is always changing it is recommended that everyone get the new seasonal vaccine every year. The strains picked for the seasonal flu vaccine are those that experts chose because they are the strains most likely to emerge in the upcoming flu season. However, experts are not always able to correctly predict the strains of flu that end up circulating, so even if you get the vaccine, it IS possible to still get the flu. But, if you get the flu vaccine and still get the actual influenza virus, you may still experience a lesser sick time and fewer symptoms.
Antibodies to these vaccines decline over the year, so the vaccine from the previous year will not cover you for the current year. Your best chance for protection against the seasonal flu is vaccination. You can find the vaccines at most doctor offices, urgent-care clinics, and from some pharmacies starting in early October. (The start of the flu season is designated as Oct. 4, each year.) You can find flu vaccines near you using the vaccine finder at http://vaccine.healthmap.org/. But make sure to call ahead to ask about availability if you are interested in a specific type of vaccine. Also, check your local health department. The vaccine most likely is available for free for your child or and at a very low cost to you. (You may want to look into getting all of your vaccines there if you think you may have to pay out of pocket.)
The seasonal flu causes about 36,000 deaths in the U.S., with about 200,000 people hospitalized every year.4 Without the yearly seasonal flu vaccine, the illness and death rates would likely be much higher. Please consider protecting yourself, your family, and your community from the seasonal flu this year. Should you come down with the flu, stay home and keep others healthy this holiday season. There’s no need pass germs on to others, and of course, there’s no need to let those germs get you! Consider vaccination this year.
References:
- United States Centers for Disease Control and Prevention (CDC). www.cdc.gov
- Iliades, Chris. “Is the New Quadrivalent Flu Shot Better?” Everyday Health. 10 Oct. 2013. http://www.everydayhealth.com
- U.S. Department of Health & Human Services. Flu.gov. 200 Independence Avenue, S.W. – Washington, D.C. 20201 www.flu.gov
- Davis, Charles Patrick and Melissa Conrad Stöppler. eMedicineHealth. “The Flu Vaccine.” 2 Sept. 2014. www.eMedicineHealth.com
- World Health Organization. www.who.int.
- United States Centers for Disease Control and Prevention. “Prevention and Control of Seasonal Influenza with Vaccines: Recommendations of the Advisory Committee on Immunization Practices (ACIP) — United States, 2014-15 Influenza Season.” MMWR63.32. 15 Aug. 2014: 691-697. Link.
- Monto, A.S., S.E. Ohmit, J.G. Petrie, et al. “Comparative Efficacy of Inactivated and Live Attenuated Influenza Vaccines.” N Engl J Med 361 Sept. 24, 2009:1260.
Photo courtesy of:
- 2014-2015 Influenza Infographic. U.S. Department of Health & Human Services. Flu.gov. 200 Independence Avenue, S.W. – Washington, D.C. 20201 www.flu.gov